|
The Frank–Read source is a mechanism that is able to produce a stream of dislocations from a pinned segment of a dislocation. The steps and ledges at the grain boundary are an important source of dislocations in the early stages of plastic deformation. Irregularities at the grain boundaries in materials can produce dislocations which propagate into the grain. 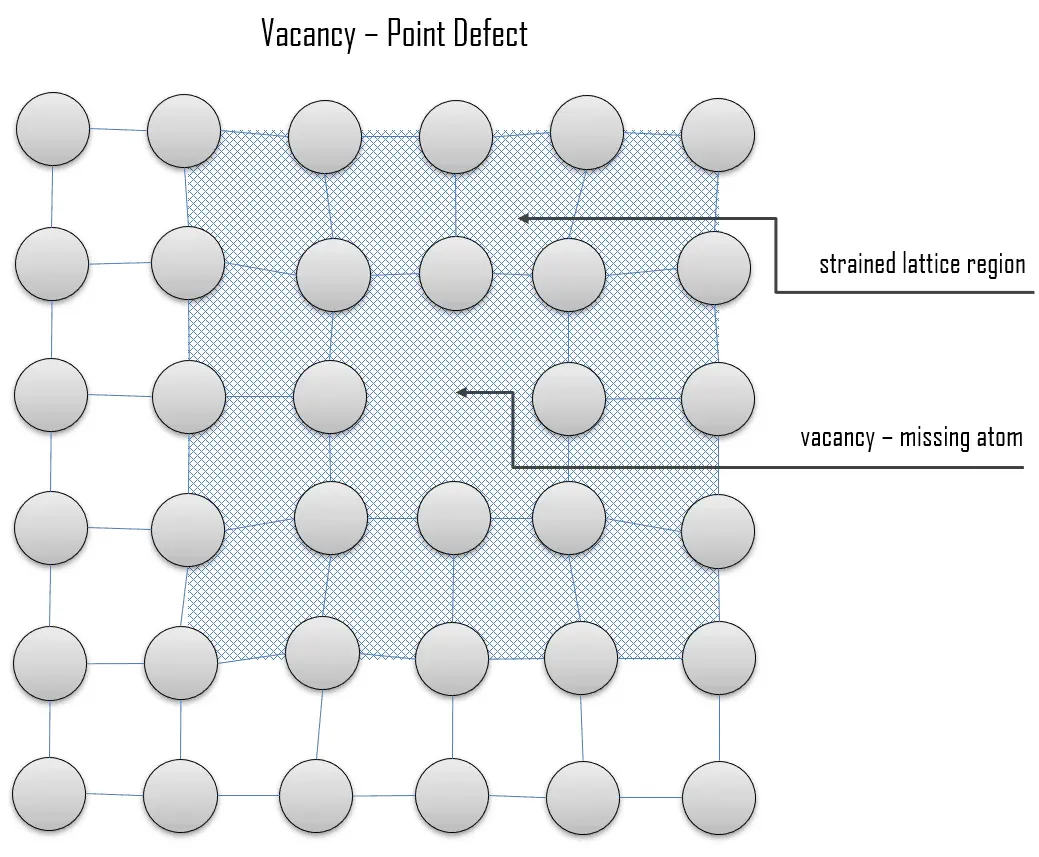
Grain boundary initiation and interface interaction are more common sources of dislocations. Therefore, in conventional deformation homogeneous nucleation requires a concentrated stress, and is very unlikely. A simplistic attempt to calculate the shear stress at which neighbouring atomic planes slip over each other in a perfect crystal suggests that, for a material with shear modulus G, we see that the required stress is 3.4 GPa, which is very close to the theoretical strength of the crystal. 
Prior to the 1930s, one of the enduring challenges of materials science was to explain plasticity in microscopic terms. The term 'dislocation' referring to a defect on the atomic scale was coined by G. The theory describing the elastic fields of the defects was originally developed by Vito Volterra in 1907. Taylor, proposed that the low stresses observed to produce plastic deformation compared to theoretical predictions at the time could be explained in terms of the theory of dislocations. In 1934, Egon Orowan, Michael Polanyi and G. This phenomenon is analogous to half of a piece of paper inserted into a stack of paper, where the defect in the stack is noticeable only at the edge of the half sheet. In such a case, the surrounding planes are not straight, but instead bend around the edge of the terminating plane so that the crystal structure is perfectly ordered on either side. 
The two main types of mobile dislocations are edge and screw dislocations.Įdge dislocations can be visualized as being caused by the termination of a plane of atoms in the middle of a crystal. Examples of sessile dislocations are the stair-rod dislocation and the Lomer–Cottrell junction. The two primary types of dislocations are sessile dislocations which are immobile and glissile dislocations which are mobile. The number and arrangement of dislocations influences many of the properties of materials. Plastic deformation of a material occurs by the creation and movement of many dislocations. A dislocation can be characterised by the distance and direction of movement it causes to atoms which is defined by the Burgers vector. A dislocation defines the boundary between slipped and unslipped regions of material and as a result, must either form a complete loop, intersect other dislocations or defects, or extend to the edges of the crystal. The crystalline order is not fully restored with a partial dislocation. The crystalline order is restored on either side of a glide dislocation but the atoms on one side have moved by one position. The movement of dislocations allow atoms to slide over each other at low stress levels and is known as glide or slip. In materials science, a dislocation or Taylor's dislocation is a linear crystallographic defect or irregularity within a crystal structure that contains an abrupt change in the arrangement of atoms. Dislocations of edge (left) and screw (right) type. For the medical term, see Joint dislocation. During deformation, progressive movement along flaws in crystals takes place in tiny steps, as bonds are broken and re-formed.For the syntactic operation, see Dislocation (syntax). Such defects affect the mechanical strength of a crystal, which determines the strength of rocks and how they deform under intense pressure. Figure 22 shows an artificial example based on an alloy used in semiconductors, where each dark line represents a strained part of the crystal. 
Minerals in deformed rocks, such as those from mountain belts, contain large numbers of line defects caused by rows of atoms that are out of place in the crystal structure. This is important because the rate of diffusion of atoms through a crystal structure can determine the speed at which processes such as weathering, or other chemical reactions (e.g. Such defects make it much easier for atoms to diffuse through the crystal structure by moving between vacant sites. Point defects may involve missing or displaced atoms in the crystal structure, giving empty sites, or vacancies. The effect of defects on the physical and chemical properties of a crystal can be out of all proportion to their size.ĭefects come in several types. Virtually all crystals contain minute imperfections or defects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
